Malaysia will be able to use the Pfizer vaccine for COVID-19 upon the expected arrival of its first batch in the country next month, following a conditional approval for the vaccine by a government regulator.
Minister Khairy Jamaluddin today announced on Twitter that the National Pharmaceutical Regulatory Agency (NPRA) has given its conditional registration for the vaccine, noting that this information was conveyed to him by Health director-general Tan Sri Dr Noor Hisham Abdullah.
“I was just informed by @DGHisham that NPRA has given conditional registration for the Pfizer vaccine. We are still waiting for a few additional info from Pfizer but this means it can be used in Malaysia. Congratulations NPRA on the quick registration,” the minister of science, technology and innovation wrote in a brief tweet.
On November 27, Prime Minister Tan Sri Muhyiddin Yassin announced that Malaysia had on November 24 signed a preliminary deal with Pfizer to buy 12.8 million doses of COVID-19 vaccine — sufficient for 6.4 million persons or 20 per cent of Malaysia’s population.
The 12.8 million doses are expected to be delivered quarterly in 2021 in installments of one million doses (first quarter of 2021), 1.7 million (second quarter), 5.8 million (third quarter), 4.3 million (fourth quarter).
The COVID-19 vaccine was co-developed by US pharmaceutical giant and listed German biotechnology firm BioNTech SE, with two doses per person required for vaccination which the companies had said has an efficacy rate of more than 90 per cent.
While the government plans to provide COVID-19 vaccination for free to Malaysians with a priority on frontliners, senior citizens and high-risk groups but would charge foreigners a fee for such vaccination, Malaysia had previously said it will only start using such vaccines if approved by the country’s NPRA.
On December 21, Health D-G Dr Noor Hisham said the NPRA had on December 15 received Pfizer’s application to register its COVID-19 vaccine with the regulator, with 11 medical experts to help look into the documents received and to help in evaluating the vaccine’s quality, safety and efficacy and with the evaluation process possibly taking 90 to 120 days.
Dr Noor Hisham had said the evaluation by NPRA was the first of five steps taken for the use of a COVID-19 vaccine in Malaysia, with the other four steps being ensuring procurement and logistics, determining priority with frontliners and the most vulnerable to be the first vaccinated, monitoring the side-effects, and evaluating whether the vaccine is effective in curbing the spread of Covid-19 infections.
On December 22, Dr Noor Hisham in a Facebook post had further explained the process for approval of use of pharmaceutical products such as COVID-19 vaccine in Malaysia, noting that it must first be evaluated by the Health Ministry’s NPRA and subsequently registered with the Drug Control Authority (DCA) before it can be marketed in Malaysia.
He had said that the timeline for 90 to 120 working days is for priority review within NPRA of the documents submitted before it makes a recommendation to the DCA, but noted that the NPRA was—”in the interest of public emergency”—committed to providing a comprehensive review that is well within the stipulated timeline or earlier while at the same time complying with all international guidelines.
“The final decision on the usage of a vaccine will only be made after receiving approval from DCA and in compliance with prevailing acts and regulations of Malaysia,” he had said then.
On December 31, the World Health Organisation (WHO) granted emergency validation to the Pfizer-BioNTech vaccine, the first of COVID-19 vaccine candidates to receive the international body’s emergency validation.
The WHO emergency validation for the Pfizer-BioNTech vaccine was expected to pave the way for regulators in countries globally to approve the import and distribution of the vaccine.
WHO had said its own experts and experts globally had reviewed data on the Pfizer-BioNTech vaccine and found that it met the mandatory criteria for “safety and efficacy” set by WHO, and that the benefits of using the vaccine to address the COVID-19 pandemic offset potential risks.
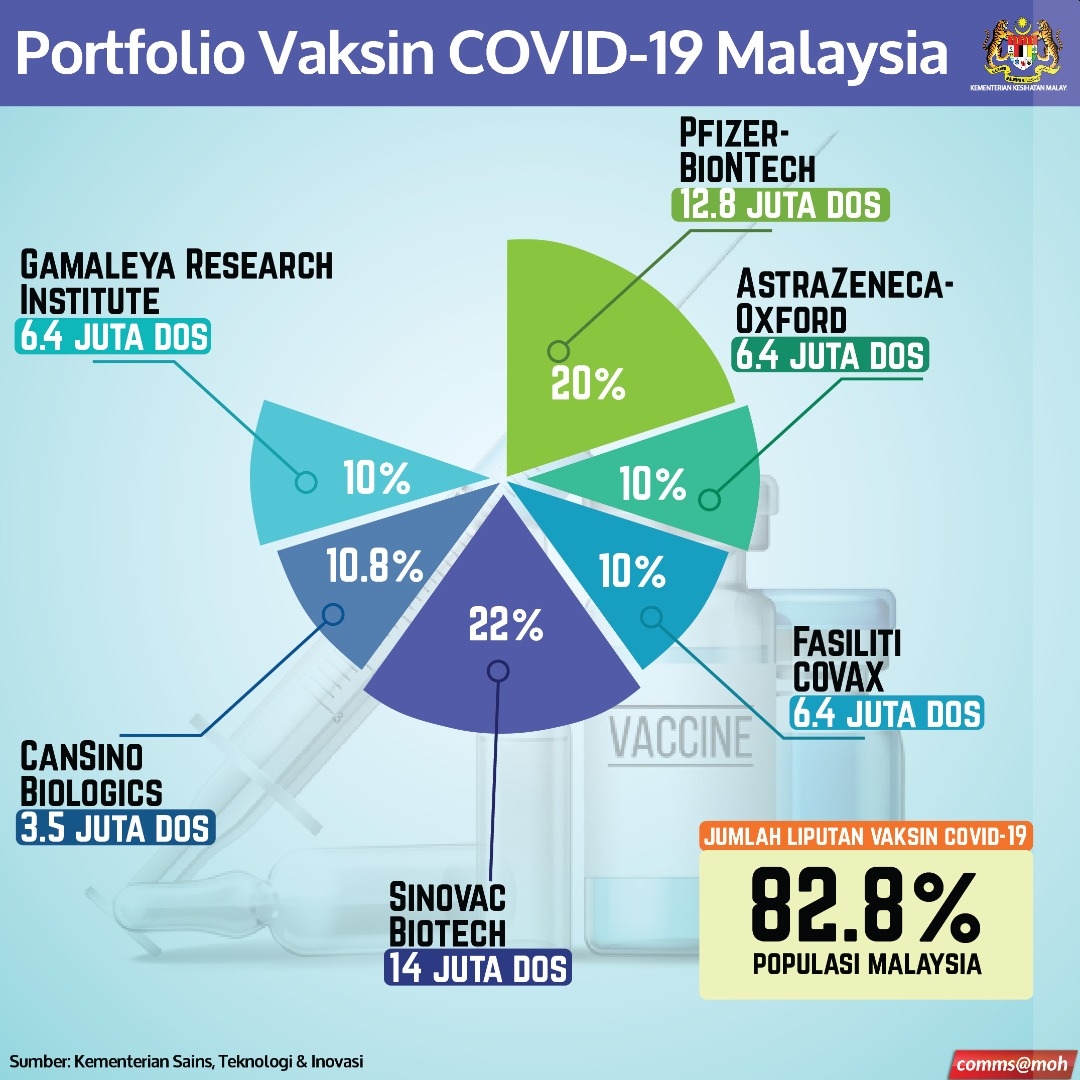
Apart from the Pfizer-BioNTech vaccine for 20 per cent of Malaysia’s population, the Malaysian government had in December 2020 also said that it had secured supply of another COVID-19 vaccine co-developed by UK-based pharmaceutical firm AstraZeneca and the University of Oxford for 6.4 million persons or another 20 per cent of Malaysia’s population, with this vaccine expected to arrive in batches between March to June 2021.
The government’s initial target was to get enough COVID-19 vaccines for 70 per cent of the population to achieve herd immunity.
As of December 22, the government estimated that it will be spending RM2.05 billion out of a RM3 billion allocation to provide free COVID-19 vaccines to 82.8 per cent or 26.5 million of the Malaysian population, subject to ongoing negotiations with two suppliers from China and a Russia supplier to cover the remaining 42.8 per cent of the 82.8 per cent estimate. — Malay Mail








