The Ministry of Health is aware of some individuals and organisations calling for the use of Ivermectin to treat COVID-19 cases in Malaysia. Health Director-General Dr Noor Hisham has posted a response on the usage of the drug and Malaysia’s attempt to evaluate its safety and efficacy.
Ivermectin only authorised for animals
As highlighted by the Health D-G previously, Ivermectin is only licensed for use for animals in Malaysia. It is primarily used as an anthelminthic drug used to treat various parasitic infestations. The National Pharmaceutical Regulatory Agency (NPRA) has not approved any products with Ivermectin as an active ingredient to be used for human consumptions.
Ivermectin not authorised for use by FDA and EMA

He added that the US Food Drug Administration (FDA) has not yet approved the use of the drug in treating or preventing COVID-19 in humans. This is also highlighted in an article on the FDA website titled Why You Should Not Use Ivermectin to Treat or Prevent COVID-19. It also warned that taking large doses of the drug is dangerous and can cause serious harm.
He quoted a placebo-controlled randomised trial in January 2021 which involved 24 non-severe COVID-19 patients. The trial didn’t manage to show a reduction in the proportion of PCR positive patients at day 7 after taking Ivermectin 400 mcg/kg single oral dose treatment. The authors of the study which was published on EClinical Medicine (Lancet) had opinionated that larger trials may be needed to understand the effects of Ivermectin.
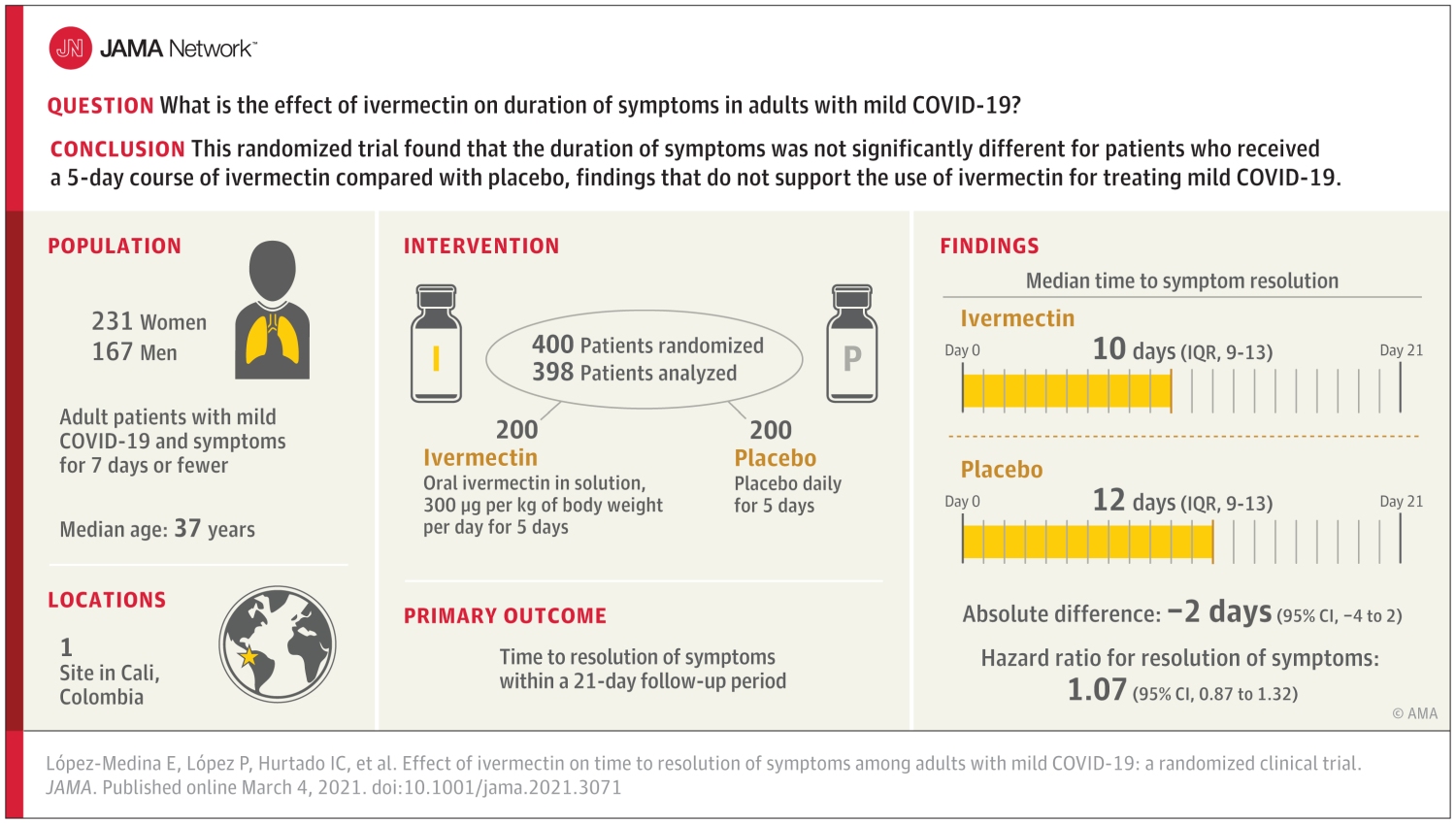
In March this year, the Journal of American Medical Association (JAMA) published a randomised-control trial involving 476 adults with mild COVID-19 disease. After a 5-day course of Ivermectin, the results showed no significant improvement in COVID-19 symptoms resolution time. While the drug may have some anti-viral and anti-inflammatory effects, the evidence is said to remain inconclusive.
In Europe, the European Medicines Agency (EMA) has advised against the use of Ivermectin for the prevention or treatment of COVID-19 outside clinical trials. According to the EMA, laboratory studies have shown that Ivermectin could block the replication of SARS-CoV-2 but at a much higher Ivermectin concentration than what is currently authorised. It added that most of the studies conducted were small and had additional limitations. The EMA concluded that the current evidence is insufficient to support the use of the drug to treat COVID-19.
Ivermectin trials in Malaysia
However, the Ministry of Health Malaysia has not completely ruled out the use of Ivermectin for COVID-19 cases just yet. It has initiated randomised clinical trials to repurpose Ivermectin and to evaluate its efficacy and safety in high-risk COVID-19 patients. The trial will enroll 500 COVID-19 patients admitted to 12 MOH hospitals. It hopes that the study will provide a clearer perspective on the clinical effectiveness of Ivermectin.
Dr Noor Hisham emphasised without the benefit of evidence from well-designed clinical trials, the Ministry of Health will not be able to endorse Ivermectin to prevent or treat COVID-19 illness. He stresses that science is the best weapon against SARS-Cov-2 virus, not circumstantial hype.
At the moment, the best prevention against COVID-19 is to get vaccinated and to maintain SOP such as wearing a face mask and social distancing. All vaccines that are currently administered in Malaysia have been approved by the NPRA after evaluating their safety and efficacy.
[ SOURCE 2, IMAGE SOURCE ]